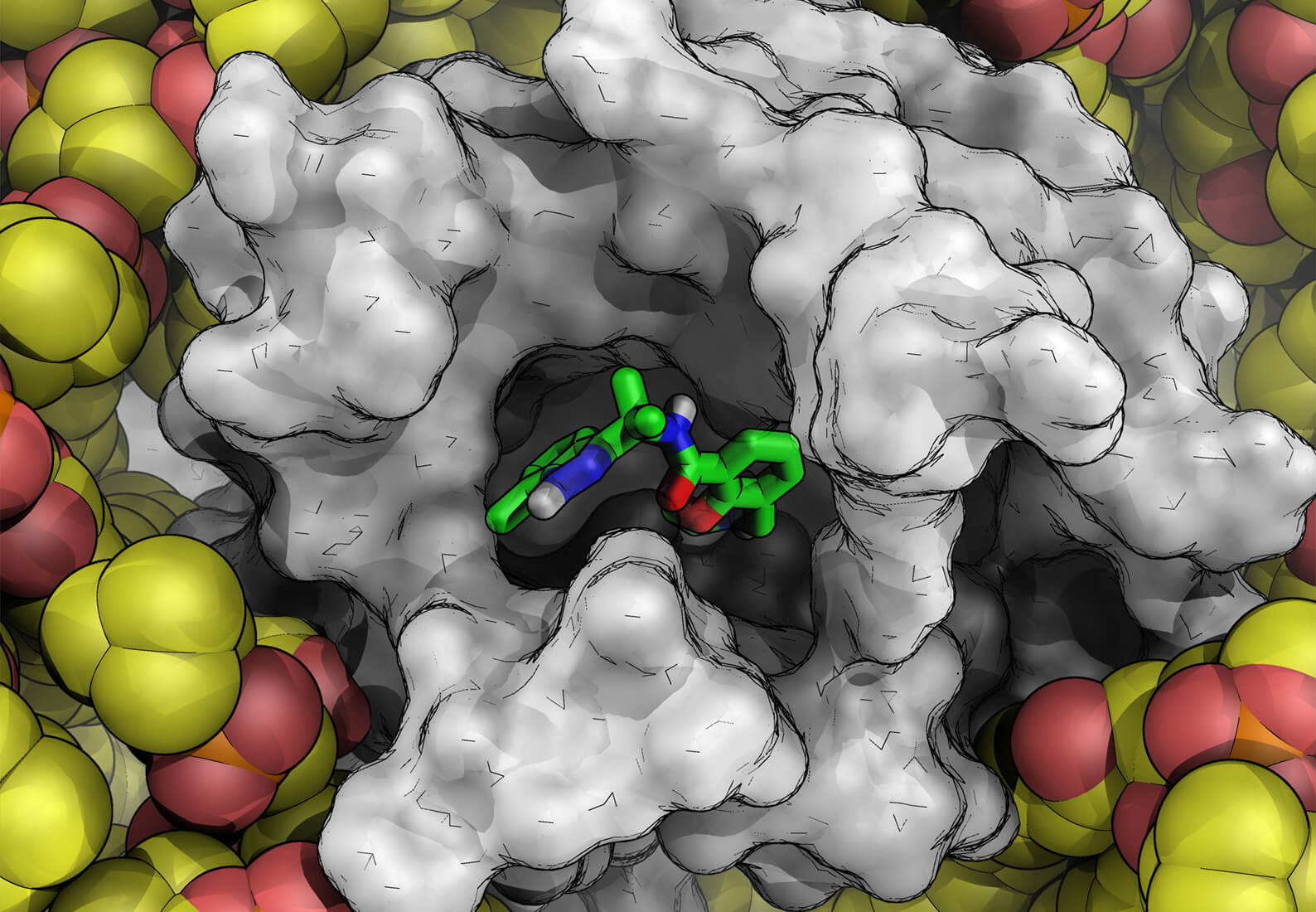
The complexity and size of drug candidates has grown in recent years as scientists pursue novel targets once considered undruggable. As a result, discovery teams are increasingly faced with the challenge of compounds demonstrating poor aqueous solubility. The solubility of a drug molecule is vital for its bioavailability. If a drug is not sufficiently soluble, it cannot be fully absorbed into circulation and will be expelled before reaching its site of action. Furthermore, poor solubility can lead to significant and extensive formulation efforts to enable efficacy and toxicology studies.
Despite its critical importance, early assessment of crystalline thermodynamic solubility continues to be elusive for drug discovery and development. Scientists from AbbVie and Schrödinger have been collaborating to explore new physics-based methods for solubility prediction. The results of this work were published in the Journal of Medicinal Chemistry and summarize the development and validation of a free energy perturbation based approach for aqueous crystalline solubility prediction.
We sat down with authors Richard Hong, Ph.D., from AbbVie and Ana Rojas, Ph.D. from Schrödinger to discuss the collaboration and findings from the study.
What motivated you to pursue this research? Why is it important to be able to assess and predict crystalline thermodynamic solubility?
Richard: Getting precise measurements of crystalline solubility in early stages of drug discovery is very important for understanding a compound’s developability challenges. Often, challenges related to crystalline solubility are not discovered until later in the drug discovery process, where significant resources have already been invested to advance a compound. Predicting crystalline solubility at an early stage not only enables the identification of potential risks but also aids chemists in prioritizing synthesis and molecular design.
Historically, which methods have you used to address this problem?
Richard: In the early stages of drug discovery, high-throughput assays are commonly employed to experimentally assess kinetic solubility. However, these solubility assays can often provide a significant overestimate of the thermodynamic solubility, sometimes more than 1000 times.
Furthermore, obtaining precise and controlled measurements of crystalline solubility is usually not feasible until later stages of drug discovery when the compound has been scaled up and thoroughly characterized. Consequently, there is limited data available for high quality crystalline solubility, especially for structural analogues. For these reasons, developing predictive machine learning models to accurately assess crystalline solubility and differentiate between structural analogues is quite challenging. While machine learning models offer the advantage of instant predictions, they do not provide the structural and energetic insights necessary to help guide the design of more soluble compounds.
What are the key findings from the study?
Ana: In the study, we applied a physics-based method that utilizes FEP+ technology to compute thermodynamic aqueous solubility (solubility FEP+). We assessed the predictive power of this approach across diverse regions of chemical space, spanning pharmaceutically relevant literature compounds and complex AbbVie compounds. Our approach achieves strong predictive and differentiating power and provides notably improved accuracy compared to state-of-the-art machine learning approaches.
Richard: This study emphasizes the importance of explicit considerations of crystal packing and energetics when evaluating solubility. For instance, traditional approaches of enhancing solubility by introducing additional extra polar groups may inadvertently reduce the solubility by strengthening hydrogen bonding interactions within the crystal. By integrating predicted or experimental crystal structures with solubility FEP+, we can get precise estimations of crystalline solubility in addition to energetic and structural design insights into how to improve solubility.
How do you anticipate the findings impacting your workflow going forward?
Richard: At AbbVie, we envision solubility FEP+, combined with Crystal Structure Prediction, to be readily implemented as part of the medicinal chemistry design funnels for programs that exhibit solubility challenges — especially for compounds that are difficult to synthesize.
By using this method, we can assess compound design ideas, and compounds showing promising solubility predictions (along with other favorable predicted properties) can be prioritized for synthesis. Furthermore, this approach not only provides a predicted value for solubility but also offers atomistic insights into how specific design ideas influence crystalline solubility. For example, it can help scientists understand whether the addition of certain substituents can likely disrupt crystal packing. Additionally, for programs that have historically faced challenges with poor crystalline solubility, this approach enables retrospective learnings regarding the structural and energetic contributions to this issue.
With future developments in Crystal Structure Prediction algorithms and the widespread availability and advancement of computing hardware, we believe that synthetic designs can be routinely profiled on a weekly basis, providing not only solubility predictions, but also energetic and structural insights into the solubility value.
What’s next for the collaboration between AbbVie and Schrödinger?
Ana: This was an extremely productive collaboration. The ingenuity of our colleagues at AbbVie when trying to understand issues with difficult compounds challenged our scientists to find ways to address these issues. I believe this model will lead us to the development of the next generation of solubility FEP+ workflows as we adjust to address the increasingly complex chemical space of drug candidates and new modalities in the field.
Richard: Through multiple brainstorming sessions with Schrödinger, we were able to identify key problem statements and generate ideas on how bespoke state-of-the-art simulations can be utilized to address them. From this, we were able to develop and implement novel workflows to tackle crystalline solubility prediction — a pertinent industry challenge. We hope that this collaboration model leads to new developments in practical in-silico solutions for unresolved scientific challenges.